ISO 9001:2015 Certified Practice
350+
Practitioners Worldwide
20+
Years of IP Excellence
15+
Jurisdictions Covered
3
Core Filing Strategies
Adamantane & Dihydrazide-dihydrazone Group Compound, represented by A2P Care Pharmaceuticals LLP
The patented compound merges the therapeutic power of adamantane and dihydrazide-dihydrazone groups, known for their efficacy in treating Type 2 diabetes, neurological disorders like Parkinson’s and Alzheimer’s, and cancer. The dihydrazide-dihydrazone ability to address anti-diabetes and hypertension gives it an edge over existing treatments.
Overview
Our patented compound contains adamantane and dihydrazide-dihydrazone groups, both of which are known for their medicinal properties, as demonstrated in numerous literature reviews. The compound holds promise in the treatment of several diseases, especially cancer, neurological disorders, and diabetes.
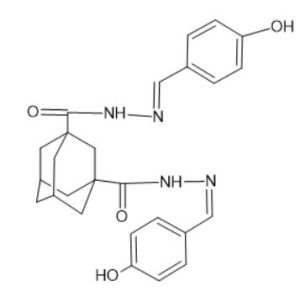
Adamantane Derivatives:
Widely recognized for their efficacy in treating neurological conditions, such as Parkinson’s disease and Alzheimer’s disease, adamantane-based compounds are expected to play a pivotal role in the pharmaceutical industry in the near future. According to existing research, adamantane’s ability to mitigate neurological complications associated with Type 2 diabetes further strengthens its therapeutic potential.
Dihydrazide-Dihydrazone Compounds:
Known for their effectiveness against hypertension and diabetes, these compounds also exhibit various other pharmacological activities. The literature highlights the specific benefits of hydrazide/hydrazone derivatives in inhibiting alpha-glucosidase, a key enzyme related to Type 2 diabetes management.
For more details about technology: Click Here
Patent Application No.: : 202221033544
PCT Patent No.: WO2023237924A1
HIPATONE
HIPATONE stands out as a revolutionary solution in the landscape of liver treatment for several compelling reasons. It harnesses the power of nature through a meticulous blend of extracts from Curcuma longa, Phyllanthus emblica, and Gymnosporea montana, each renowned for their hepatoprotective and medicinal properties. Not only does it address the symptoms of liver dysfunction like jaundice, but it also targets the underlying causes with its potent anti-inflammatory and antioxidant properties. By reducing liver inflammation and protecting liver cells from oxidative damage, Hipatone not only alleviates current symptoms but also prevents further deterioration of liver function.
The liver is the principal organ that is capable of converting drugs into forms that can be readily eliminated from the body. Given the diversity in use today and the complex burden they impose upon the liver, it is not surprising that a broad spectrum of adverse drug effects on liver functions and structures has been documented. The reactions range from mild and transient changes in the results of liver function tests to complete liver failure with death of the host. Many drugs may affect the liver adversely in more than one way, ascited below in several listings. The use of the following drugs requires careful monitoring of their effects on the liver during the entire course of treatment CLINICAL INDICATIONS LIVER DYSFUNCTION & JAUNDICE DETOXIFY LIVER THROUGH NATURAL SOURCE OF COMPOUNDS. This list is just a general guideline. Many drugs affect the liver to one degree or another and we can’t list all of them here; new drugs are always being approved for general use. “Liver Malfunction-Jaundice” – novel research composition signifies that HIPATONE®isanalytically and clinically proven safeand effective
For more details about technology: Click Here
Patent Application No.: 153/MUM/2011 (NBA Approval awaited)
US Patent No.: US8431167B2
CARDIMODE
CARDIMODE stands out as a beacon of hope in the realm of cardiovascular health, offering a compelling solution backed by science and nature. With its unique blend of botanical ingredients, including Terminalia arjuna, Camellia sinensis, and Trikatu, this proprietary herbal medicine presents a promising avenue for individuals seeking to manage their cholesterol levels and safeguard their heart health. Through its targeted action on LDL (bad) cholesterol reduction and HDL (good) cholesterol elevation, this herbal supplement offers a holistic approach to cholesterol management, addressing a key risk factor for heart disease.
LITERATURE: UNDERSTANDING HEART ATTACK
A heart attack, or myocardial infarction, occurs when a blood vessel supplying the heart muscle is suddenly blocked by a blood clot. This blockage typically arises from the accumulation of cholesterol and other fatty substances within the blood vessel, forming plaque. Over time, plaque buildup narrows and hardens the arteries, a condition known as atherosclerosis. Reduced blood flow to the heart can lead to symptoms such as chest pain (angina) and, if left untreated, permanent damage to the heart muscle.
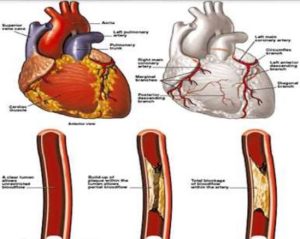
CARDIMODE Heart is vulnerable to a myriad of risk factors that contribute to heart disease. These include smoking, high cholesterol, high blood pressure, diabetes, obesity, and a sedentary lifestyle, collectively posing a significant threat to cardiovascular health. Plaque accumulation in the arteries, often triggered by elevated cholesterol levels and other risk factors, can culminate in partial or complete blockages, precipitating heart attacks. Additional risk factors encompass genetic predisposition, metabolic syndrome, heavy alcohol consumption, stress, sleep apnea, and certain medical conditions like preeclampsia. Embracing a healthy lifestyle, characterized by regular exercise, a balanced diet, smoking cessation, and effective stress management, is imperative in averting heart disease and mitigating the-likelihood of heartattacks.
For more details about technology: Click Here
Patent Application Number: WO2013021302A
Masktag: Accessory Device For Face Coverings
Accessory Device for Face Coverings
The described invention pertains to an accessory device designed to address various issues associated with the use of face coverings, surgical masks, and filtering facepieces, especially in the context of the COVID-19 pandemic. Here’s an overview of the problem it aims to solve and the features it offers:
Problem Statement:
The widespread use of face coverings and masks in response to health emergencies, such as the COVID-19 pandemic, has highlighted several challenges and shortcomings associated with these protective devices. Some of the key issues include:
1. Difficulty in recognizing one’s mask after temporary removal, which poses a risk of using a contaminated mask.
2. Impaired recognizability of the wearer due to the partial coverage of the face by the mask, particularly exacerbated when wearing additional accessories like glasses or hats.
3. Accumulation of unpleasant odors inside the face covering, primarily caused by the user’s breath.
4. Build-up of microbial and viral load within the face covering due to prolonged use and favorable microclimatic conditions, potentially leading to health risks.
CARDIMODE Heart is vulnerable to a myriad of risk factors that contribute to heart disease. These include smoking, high cholesterol, high blood pressure, diabetes, obesity, and a sedentary lifestyle, collectively posing a significant threat to cardiovascular health. Plaque accumulation in the arteries, often triggered by elevated cholesterol levels and other risk factors, can culminate in partial or complete blockages, precipitating heart attacks. Additional risk factors encompass genetic predisposition, metabolic syndrome, heavy alcohol consumption, stress, sleep apnea, and certain medical conditions like preeclampsia. Embracing a healthy lifestyle, characterized by regular exercise, a balanced diet, smoking cessation, and effective stress management, is imperative in averting heart disease and mitigating the-likelihood of heartattacks.
For more details about technology: Click Here
Patent Application Number: WO2013021302A
Patent Protected Modular Urinal for Bed Ridden People
About Us
IIPRD is representing Peeshcute in an IP Licensing mandate for a technology which Empowers Convenience and Independence for those with limited mobility issues.
Peeschute’s modular urinal tech has been carefully designed considering factors like non-invasiveness, ease of use, hygiene, sustainability, reduced dependability and reduced risk of infections.

- Established in 2018, Peeschute is a renowned brand specializing in developing innovative solutions for sanitation.
- Peeschute has developed urinal products for travel, healthcare/homecare and outdoor spaces.
- Peeschute’s flagship product includes pocket sized disposable urine bags which solidifies up to 850 ml of urine instantly to keep it leak proof, odorless and hygienic.
- Peeschute’s products have been accoladed with national and international attention including media, organizational collaborations with government and non-government bodies.
Patent Application No: 202221026004
Registrations Design Number: 373040-001 and 373041-001
For more details about technology: Click Here
“Resoil” presented by Envit Ltd.
ReSoil® is an internationally patented, groundbreaking remediation solution that enables closed-loop removal of heavy metals from contaminated soils through a zero-emission process with no wastewater, while preserving soil as a natural substrate.
• Soil contamination with heavy metals is a global problem.
• Heavy metals include toxic metals such as lead (Pb), cadmium (Cd), copper (Cu) and zinc (Zn) as well as toxic metalloids such as arsenic (As) and antimony (Sb).
• Soils absorb decades of pollution from industry, intensive agriculture, traffic, waste, etc. Heavy metals are not degradable and remain in the soil.
• According to a recent conservative estimate, there are 2.8 million potentially contaminated sites in the EU (EEA, 2023).
• In China, 1/6 of the total cultivated area is contaminated with heavy metals (Yao et al., Proc Env Sci 2012, 16, 722-729).
• In New York, 71% of garden soils exceed the limits for lead and arsenic (Chenget al., Soil Sci 2015, 180, 167-174).
• Arsenic and lead are the 1st and 2nd most dangerous environmental pollutants
(ATSDR 2017).Lead distribution in surface soil of the conterminous United States (USGS 2014).
• Arsenic is highly toxic, long-term exposure can cause cancer.
• Lead is a neurotoxin; in the USA, the average lead-related loss of cognitive ability was 2.6 IQ points per person as of
2015 (McFarland et al. PNAS 2022, 119, e2118631119
For more details about technology: Click Here
Patent Application Number: WO2022184903
Machine to Dispose off used Sanitary napkins chemically
Current methods of disposal and associated danger
1. Flushing and burying
• leads to blockages, plumbing problems
• outer layer of sanitary napkin is non-degradable
• collection of napkins in heaps block the pipeline
• pollute streams and rivers
• causes hormonal changes in wildlife – enable harmful organisms in the food chain
2. Incineration
• Harmful gas emissions: dioxin, furan, nitrogen oxides, sulphur dioxide, hydrochloric acid, mercury
• Ash produced contain heavy metals like lead, cadmium, copper, zinc
• Dioxins: cancer, immune system damage, reproductive and developmental problems.
• Halogenated hydrocarbons, acid gases – impair lung function.
Objectives of the new machine
• Disposal of sanitary napkins properly, thereby wiping out the risks involved in flushing, burying, and incineration.
• Reduce pollution and health hazards of conventional methods of sanitary waste disposal.
• Reduce cost for proper disposal of sanitary napkins, tampons, baby diapers and under pads.
• Simplify the process of sanitary waste disposal.
• Help in solving greatest problem people face daily.
Key Points of the machine
• One machine- can be used for sanitary napkins, tampons, baby diapers and under pads. (If a method to choose the option while
using the machine is implemented)
• Can be manufactured both – domestic (for households) and industrial size (waste disposal plants/ panchayats)
Patent Application Number: 298227
For more details about technology: Click Here
DIABETES CARE – MEDICAL DEVICE FOR TISSUE OXIMETRY
The Opportunity
Blackhawk Technologies, LLC is exclusively representing HyperMed Imaging Inc. (the“Company”) to monetize its’ assets related to a Medical Device for Tissue Oximetry for Diabetes Care. The offering includes an FDA cleared product used by clinicians and backed by 24 patent families comprising of 62 total assets.
The Company’s pioneering technology has been in service since 2006 and has been validated in third-party published papers that assessed oxyhemoglobin, deoxyhemoglobin, and oxyhemoglobin saturation in superficial tissue for diabetic patients. The Company’s FDA 510(k) cleared product, Hyper View™, quickly determines levels of oxyhemoglobin, deoxyhemoglobin and oxygen saturation in superficial tissue in a small and portable configuration.
The Company seeks a strategic acquirer/licensee with both resources and channels to fully commercialize Company’s IP assets.
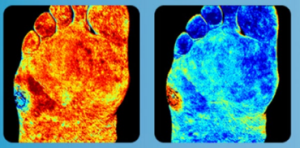
The Unmet Medical Need
Most medical exams need quick and accurate determination of superficial tissue oxygenation. For example, complications in diabetes and/or peripheral arterial disease increases risk of lower limb ischemia, chronic wounds, amputation or even death.
• Over 420 million people worldwide (1 in 11 people) have diabetes and the number is increasing rapidly.
• More than 29 million Americans have diabetes. Another 86 million have prediabetes, which is 1 in 3 US adults.
• 8.5 million Americans have peripheral arterial disease and among them nearly 2 million people in the USA are living with limb loss.
• The main causes are vascular disease (54%), including diabetes and peripheral arterial disease.
• Approximately 185,000 amputations occur in the United States each year, costing over $8 billion.
• People with diabetes are at greater risk for severe peripheral arterial disease and are five times more likely to require an amputation.
• It is estimated that a low
Hyperspectral Imaging Benefits
• Non-contact color-coded images depicting concentrations of oxyhemoglobin & deoxyhemoglobin, and oxygen saturation without injectable contrast agents.
• Readings are predictive of wound healing and can be used in screening for peripheral vascular disease.
• Software tools allow clinicians to analyze specified areas within the image to localize ischemic tissue.
• Offers improved patient care with speed, ease of use, and clinical efficiency without injectable contrast or physical contact of the device with the patient.
• Safe, using visible light and spectrometer imaging to quantify light absorption in hemoglobin molecules for rapid analysis without leaving the exam room.
For more details about technology: Click Here
IONOJET TM
Has developed a proprietary device (“IonojetTM”) that generates nitric oxide (“NO”) from atmospheric air in the form of a high-energy plasma/NO stream and delivers it to targeted locations of the body.
The plasma/NO stream has been shown in investigations to date:
(i)to be non-toxic
(ii) to generate NO activity up to 3 cm below the skin
(iii) to stimulate sustained biological activity in tissue for up to an hour after delivery of the therapy, and
(iv) to penetrate and disrupt biofilm

Has identified over 40 target indications in seven clinical areas based on NO’s recognized ability to:
- Increase blood flow
- Destroy pathogens (bacterial, viral and fungal)
- Regenerate tissue
- Control pain and inflammation
Has completed a dose-ranging feasibility clinical trial in the U.S. for initial target indication (Diabetic Foot Ulcers, “DFU”) to identify optimal dose & demonstrate preliminary safety/efficacy data for the device.
- Has completed PoC studies in certain target indications
- Intends to initiate PoC studies in a number of additional indications
For more details about technology: Click Here
Nutrition Innovation In Singapore
White refined sugar is almost pure sucrose. This sucrose is consistently shown in research to be quickly absorbed into the blood stream causing rises in blood sugar levels. This in turn has contributed to the global obesity and diabetes crisis.
The Nucane™ specification of raw sugar is scientifically tested to ensure that this absorption is reduced.What the unique Nucane™ process achieves is to retain at a high level the natural organics, antioxidants and polyphenols that exist in raw sugar cane, delivering a carbohydrate that is less processed, less refined, and which has been demonstrated scientifically to reduce the metabolism of sugar into the blood stream when consumed.

This means that it is a low glycemic sugar when tested to the internationally agreed ISO 26442:2010 method.Just as importantly, our technology system ensures that Nucane™ is precise and consistent at industrial scale in primary sugar mills around the world. By eliminating the refining process and applying the Nucane technology at sugar mills, Nucane™ is a less processed, less refined, high in polyphenols specification of raw sugar which has been tested to be low glycemic to international standards.
For more details about technology: Click here
Prosthetic Devices Coated With Heated Cross-Linked Fibrin
Invention relates to methods of coating the surface of prosthetic devices with fibrin and drying the fibrin-coated prosthetic devices at moderately-high temperatures for extended periods of time under low atmospheric pressure to obtain prosthetic devices coated with stable cross-linked fibrin capable of binding cells and thereby capable of integrating into tissues.
Patent Application Number: WO2010/131249
For more details about technology: Click here

