Introduction The smartphone which is used today is the result of thousands of technologies which…
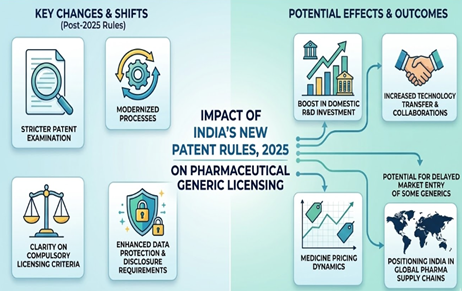
Compulsory Licensing in India Amidst the COVID 19 Pandemic: Will India Consider it?
The COVID-19 pandemic has truly exposed the frailty of our infrastructural systems. Innovation has taken a front seat and the world is keenly awaiting the public distribution of COVID-19 vaccines which would help restore normalcy. Among the numerous factors that were taken into consideration by global experts with respect to the COVID 19 vaccines, accessibility and distribution issues have taken the forefront. This, along with other factors concerning patent legislation, have put developing nations in a precarious position, pushing them to be calculative of their infrastructure, economic conditions and to evaluate their channels to mass manufacture the vaccines while deliberating the issues associated with the patentability of the COVID-19 vaccines. Before exploring the various nuances of compulsory licensing, it is important to understand the benefits of a pharmaceutical patent. A pharmaceutical patent would allow pharmaceutical conglomerates to acquire royalties, protect the innovation behind the vaccine from commercial exploitation and monopolize the invention to a very large extent. Additionally, it would receive a protection of 20 years that would be provided by the WTO members since the filing date of the patent under the TRIPS agreement. Pharmaceutical patenting would fuel innovation and R&D while ensuring that the inventors behind the product are incentivized. This is essential in order to promote growth in the pharmaceutical industry, or any sector for that matter which would in turn play a pivotal role in contributing to the economic growth of a country.
Compulsory licensing is closely related to pharmaceutical patents. It may be defined as the scenario wherein an authorization is granted by the Controller General of Patents to a third party to use, make or sell a patented product/process. Under compulsory licensing, the permission of the patent owner to make or sell the patented product/process is not necessary. Chapter XVI of the Indian Patent Act, 1970 deals with compulsory licensing. Under this chapter (particularly sections 84 and 92), various criterions are enlisted that has to be satisfied in order to grant a compulsory license. Section 84 lays down the criterions as follows:
“At any time after the expiration of three years from the date of the 170 [grant] of a patent, any person interested may make an application to the Controller for grant of compulsory licence on patent on any of the following grounds, namely: –
(a) that the reasonable requirements of the public with respect to the patented invention have not been satisfied, or(b) that the patented invention is not available to the public at a reasonably affordable price, or(c) that the patented invention is not worked in the territory of India.”
Moreover, section 92 emphasizes on criterion that are related to extra ordinary circumstances as follows:
“Notwithstanding anything contained in sub-section (2), where the Controller is satisfied on consideration of the application referred to in clause (i) of sub-section (1) that it is necessary in-
- a circumstance of national emergency; or
- a circumstance of extreme urgency;
- a case of public non-commercial use,”
In sum, it can be said that the Controller General grants an exclusive permission to use, make or sell the patented product/process under extraordinary circumstances which may include public need (instances wherein the patented product/process is not easily accessible to the general public), national emergency, extreme urgency or if the patented invention is not worked in the territory of India. Compulsory licenses have been invoked in the past. A lifesaving drug called Nexavar which was used to treat liver and kidney cancer, was incredibly expensive and unaffordable by many in India. It was speculated that a month’s dosage costed up to 2.8 Lakh. In 2012, the patent office granted a compulsory license to Natco Pharma for manufacturing Nexavar. Consequently, Nexavar was available to the general public at an affordable price of Rs. 9000.
With the onset of the COVID 19 pandemic, several drugs have become inaccessible for many due to exorbitant pricing. It would right to note that the COVID 19 pandemic is indeed a national emergency and it is indeed a matter of extreme urgency. In that light, it is speculated that the Government would grant compulsory licenses for COVID related medical products. Recently, pharmaceutical conglomerate Gilead had decided to sign a non-exclusive voluntary licensing agreement with pharmaceutical manufacturers in India (including Dr Reddy’s Laboratories and Cipla Ltd.) for the manufacturing of remdesivir in order to remove any barriers that may impede the accessibility of availing the drug in India. However, unlike compulsory licensing, the licensee here (Gilead) had voluntarily decided to share the technology behind the medicine until the WHO declares the end of the pandemic.
Over the last year, several National and International efforts have been made to remove any barriers that may affect the accessibility to COVID related medical products. This included the WTO TRIPS waiver wherein India was a co-sponsor along with South Africa. In the month of October 2020, India and South Africa submitted a joint proposal seeking the waiver of IP rights and patents under provisions of TRIPS Agreement (more specifically, sections 1, 4,5 & 7 of Part II) for COVID-19 medical supplies. The primary objective of the waiver is to ensure that there is no delay in accessibility of the vaccine due to stringent patent laws concerning the COVID 19 vaccine. However, most developed countries opposed the same and opined that this would tarnish the objectives of granting patents, its principles and that it would “send the wrong message to industry investors”.
It is pertinent to note that although India is a co-sponsor of the TRIPS waiver, the Indian Government has constantly inhibited compulsory licensing as an option amidst the pandemic, which is in contrariety with the essence of the TRIPS waiver. Moreover, during the hearing of a Suo moto case related to the pandemic in April 2021, the Supreme Court did enquire regarding compulsory licensing to the Centre especially with regards to COVID related drugs such as Remdesivir and Favipiravir. However, no satisfactory response was obtained and the final decision regarding compulsory licensing of the drugs was left to the Centre. Nevertheless, there is hope. Recently, in July 2021, a bipartisan parliamentary committee on commerce has suggested to invoke compulsory licensing on all COVID related medical products. This was published in the report titled ‘Review of Intellectual Property Rights Regime in India’. The panel suggested that granting compulsory license would help amp up the manufacturing of COVID related medical products, including COVID vaccines amidst a ‘National health emergency’. The Centre is yet to devise a response for the same. While millions across all walks of life are impacted by the pandemic, it would be prudent for the Government to invoke compulsory licensing at the earliest possible. This would expedite the manufacturing process of several drugs, thus increasing accessibility. In sum, it would be right to say that invoking compulsory licensing would be imperative in order to battle the pandemic effectively.
Author: Sanjana, a BBA LLB student of Symbiosis Law School (Hyderabad), in case of any queries please contact/write back to IIPRD at [email protected].