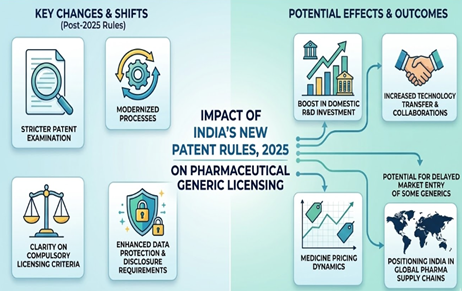
Introduction The smartphone which is used today is the result of thousands of technologies which…
Premas Biotech and Oravax enter into licensing deal for commercialization of VLP injectable vaccine technology in India
Recently, Oravax Medical Inc., (“Oravax” has entered into a licensing agreement with Premas Biotech to make available for commercialization of Oravax’s virus-like particle (VLP) vaccine technology. Oravax is a joint venture between Oramed Pharmaceuticals Inc., a US-based company (the majority shareholder), and Gurugram, India-based life sciences company, Premas Biotech (“Premas”). While the terms of the license agreement have not been disclosed, Oravax will be entitled to royalties upon commercialization of this vaccine in India.
Under this agreement, Oravax has out-licensed certain rights in the territory of India to Premas Biotech. The VLP platform technology can be delivered either via injection or orally, and under this license agreement, Oravax has licensed to Premas the right to develop an injectable version of its VLP technology with an aim to address the urgent need for a vaccine that is effective against the Delta variant. Premas plans to manufacture, test, and potentially commercialize the injectable version of the VLP vaccine in India.
The VLP vaccine is being tested in preclinical studies against COVID-19 variants including the Delta variant. The VLP vaccine technology is highly scalable with a low cost of goods and easily transferable. The oral VLP COVID-19 vaccine is being developed for use both as a standalone vaccine as well as a booster for people who have been previously vaccinated for COVID-19.
Author: Arindam Purkayastha, a Patent Attorney at IIPRD, in case of any queries please contact/write back to us at [email protected].